Electron Dot Structure Of N2
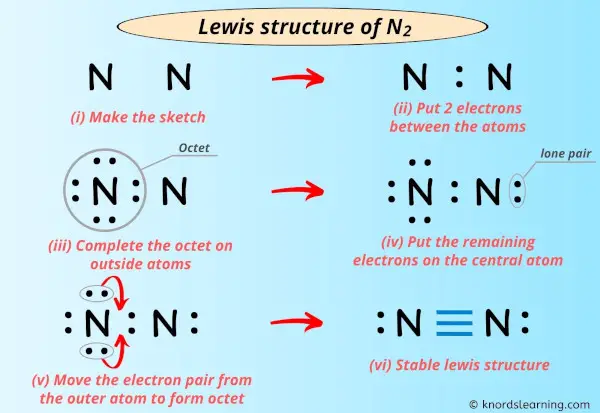
I'yard super excited to teach y'all the lewis construction of N2 in just half-dozen simple steps.
Infact, I've also given the pace-by-step images for drawing the lewis dot structure of N2 molecule.
So, if y'all are ready to go with these half dozen simple steps, then let's dive right into information technology!
Lewis structure of N2 (nitrogen gas) contains one triple bond between both the Nitrogen (Due north) atoms. And both the Nitrogen atoms have i lone pair on it.
Let's draw and empathize this lewis dot construction step by step.
(Note: Take a pen and newspaper with you and try to draw this lewis structure along with me. I am sure you will definitely learn how to draw lewis structure of N2).
6 Steps to Draw the Lewis Structure of N2
Pace #ane: Calculate the total number of valence electrons
Here, the given molecule is N2 (Nitrogen gas). In order to depict the lewis structure of N2, first of all you have to find the full number of valence electrons present in the N2 molecule.
(Valence electrons are the number of electrons present in the outermost shell of an atom).
So, let's calculate this first.
Adding of valence electrons in N2
- For Nitrogen:
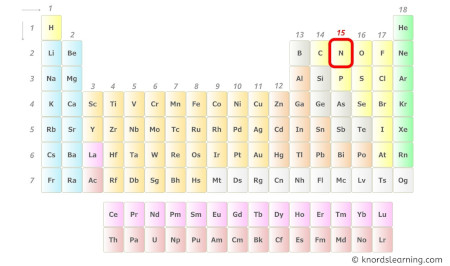
Nitrogen is a group 15 element on the periodic table.
Hence, the valence electrons nowadays in nitrogen is five (come across below prototype).
Hence, full number of Valence electrons in N2 molecule = 5(two) = ten
Step #ii: Select the center atom
While selecting the cantlet, you have to put the to the lowest degree electronegative atom at the center.
But here in the N2 molecule, both the atoms are aforementioned. So you tin consider any of the atoms as a center atom.

So, let'due south presume that the nitrogen which is on the right side is the cardinal cantlet.
Step #three: Put two electrons between the atoms to represent a chemical bond
Now in the in a higher place sketch of N2 molecule, put the two electrons (i.due east electron pair) between both the nitrogen atoms to represent a chemical bond between them.

These pair of electrons present betwixt the Nitrogen (N) atoms form a chemical bond, which bonds both the nitrogen atoms with each other in a N2 molecule.
Pace #4: Complete the octet (or duplet) on exterior atom. If the valence electrons are left, then put the valence electrons pair on the central cantlet
Don't worry, I'll explain!
In the Lewis structure of N2, we have just assumed the right side nitrogen atom equally a central cantlet so the left side nitrogen atom is an outer atom.
Then now, nosotros have to complete the octet on this left side nitrogen cantlet.
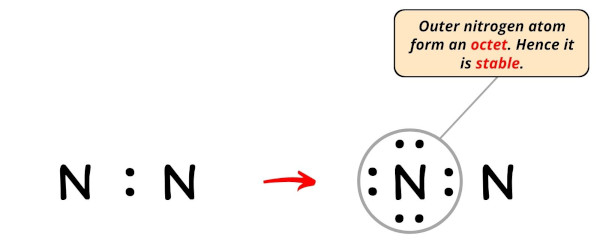
Now, yous can see in the above image that the outer nitrogen atom forms an octet.
Besides, only eight valence electrons of N2 molecule are used in the to a higher place construction.
Simply there are full 10 valence electrons in N2 molecule (as calculated in stride #1).
So the number of electrons left to be kept on the central atom = x – 8 = 2.
So let's keep these two electrons (i.east electron pair) on the central atom (i.e right side nitrogen atom).
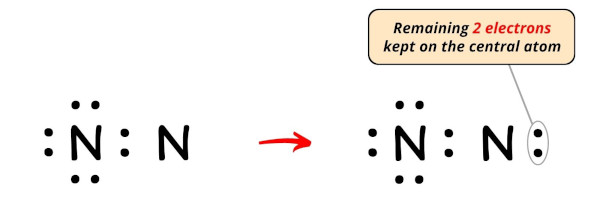
Now, permit'southward movement to the next footstep.
Footstep #5: Check whether the primal atom has octet or not. If it does not accept an octet, then motion the electron pair from the outer cantlet to form a double bond or triple bond
In this step, we have to cheque whether the central atom (i.e right side nitrogen atom) has an octet or non.
In simple words, we take to cheque whether this Nitrogen (Due north) cantlet is having 8 electrons or not.
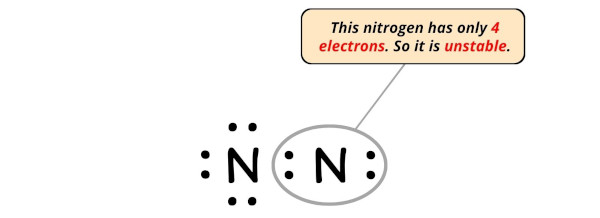
As yous can run into from the higher up epitome, this right side nitrogen cantlet has only iv electrons. So it does not fulfill the octet rule.
Now, in order to fulfill the octet of this nitrogen atom, we have to motility the electron pair from the outer atom (i.due east left side nitrogen atom) to form a double bond.
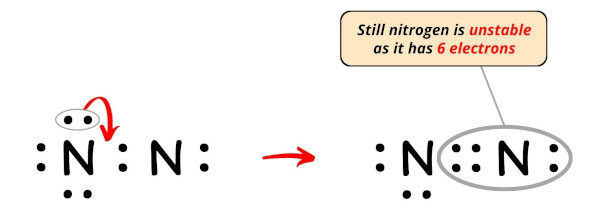
Still, the octet of nitrogen cantlet is non fulfilled as it has only vi electrons.
So once again moving the electron pair from the outer nitrogen atom, we will become the following structure.
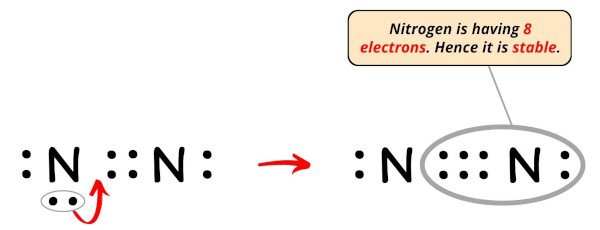
Now you lot tin can run into from the above image that the nitrogen atom has 8 electrons. So it fulfills the octet dominion and it is stable.
Step #6: Final step – Check the stability of lewis construction by computing the formal charge on each atom
Now, you have come up to the final step and here you take to check the formal accuse on the nitrogen atoms (N).
For that, you need to remember the formula of formal accuse;
Formal charge = Valence electrons – Nonbonding electrons – (Bonding electrons)/2
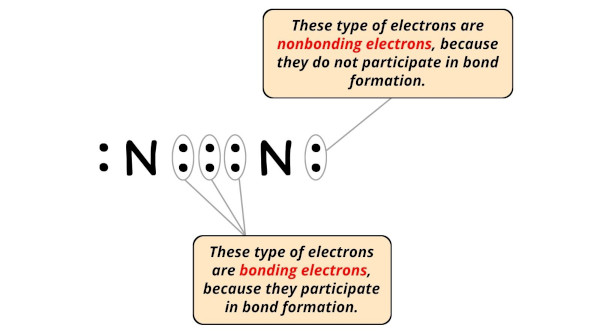
- For Nitrogen:
Valence electrons = 5 (equally it is in group 15)
Nonbonding electrons = ii
Bonding electrons = 6
| Formal charge | = | Valence electrons | – | Nonbonding electrons | – | (Bonding electrons)/two | ||
| Due north | = | 5 | – | 2 | – | 6/two | = | 0 |
So you lot can see above that the formal accuse on both the nitrogen atoms are "zero".
Hence, there will not exist any change in the in a higher place structure and the above lewis structure of N2 is the final stable construction only.
Each electron pair (:) in the lewis dot structure of N2 represents the unmarried bond ( | ). So the above lewis dot construction of N2 can likewise be represented as shown beneath.

Related lewis structures for your exercise:
Lewis construction of O2
Lewis construction of CO
Lewis structure of CH4
Lewis structure of BF3
Lewis structure of NO3 –
Electron Dot Structure Of N2,
Source: https://knordslearning.com/lewis-structure-of-n2/
Posted by: creedeneas1998.blogspot.com


0 Response to "Electron Dot Structure Of N2"
Post a Comment